Molecular Diagnostic Techniques of Infectious Diseases
An Overview
DOI:
https://doi.org/10.21141/PJP.2025.11Keywords:
molecular techniques, real-time PCR, isothermal amplification, next-generation sequencing, metagenomics, microarray, CRISPR-infectious-disease diagnosticsAbstract
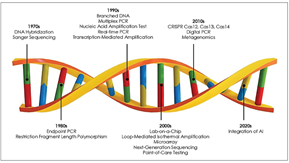
Recent advancements in molecular techniques such as real-time PCR, isothermal amplification, next-generation sequencing, metagenomics, microarray, and CRISPR-based infectious disease diagnostics have significantly evolved and improved over the past years. This overview will explore the innovations that have shaped the molecular diagnostics workflow, as well as the progress made in these emerging techniques. Additionally, it will address existing gaps, unmet needs, and potential future directions for further enhancing diagnostic capabilities in the field.
Downloads
References
World Health Organization. Pandemic and epidemic diseases: 2017 in retrospect. https://www.emro.who.int/pandemic-epidemic-diseases/information-resources/ped-2017-in-retrospect.html. Accessed July 3, 2025.
Lai CC, Shih TP, Ko WC, Tang HJ, Hsueh PR. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Int J Antimicrob Agents. 2020;55(3):105924. https://pubmed.ncbi.nlm.nih.gov/32081636 https://pmc.ncbi.nlm.nih.gov/articles/PMC7127800 https://doi.org/10.1016/j.ijantimicag.2020.105924 DOI: https://doi.org/10.1016/j.ijantimicag.2020.105924
Schmitz JE, Stratton CW, Persing DH, Tang YW. Forty years of molecular diagnostics for infectious diseases. J Clin Microbiol. 2022;60(10):e0244621. https://pubmed.ncbi.nlm.nih.gov/ 35852340 https://pmc.ncbi.nlm.nih.gov/articles/PMC9580468 https://doi.org/10.1128/jcm.02446-21 DOI: https://doi.org/10.1128/jcm.02446-21
Omar RF, Boissinot M, Huletsky A, Bergeron MG. Tackling infectious diseases with rapid molecular diagnosis and innovative prevention. Infect Dis Rep. 2024;16(2):216-27. https://pubmed.ncbi.nlm.nih.gov/38525764 https://pmc.ncbi.nlm.nih.gov/articles/PMC10961803 https://doi.org/10.3390/idr16020017 DOI: https://doi.org/10.3390/idr16020017
Liu Q, Jin X, Cheng J, Zhou H, Zhang Y, Dai Y. Advances in the application of molecular diagnostic techniques for the detection of infectious disease pathogens. Mol Med Rep. 2023;27(5):104. https://pubmed.ncbi.nlm.nih.gov/37026505 https://pmc.ncbi.nlm.nih.gov/articles/PMC10086565 https://doi.org/10.3892/mmr.2023.12991 DOI: https://doi.org/10.3892/mmr.2023.12991
Muldrew KL. Molecular diagnostics of infectious diseases. Curr Opin Pediatr. 2009;21(1):102-11. https://pubmed.ncbi.nlm.nih.gov/19242246 https://doi.org/10.1097/MOP.0b013e328320d87e DOI: https://doi.org/10.1097/MOP.0b013e328320d87e
Shin JH. Nucleic acid extraction techniques. In: Advanced techniques in diagnostic microbiology. Springer; 2012:209-25. https://doi.org/10.1007/978-1-4614-3970-7_11 DOI: https://doi.org/10.1007/978-1-4614-3970-7_11
Powell EA, Babady NE. Digital PCR in the clinical microbiology laboratory: another tool on the molecular horizon. Clin Microbiol Newsl. 2018;40(4):27-32. https://pubmed.ncbi.nlm.nih.gov/15872277 https://pmc.ncbi.nlm.nih.gov/articles/PMC1153761 https://doi.org/10.1128/JCM.43.5.2435-2440.2005 DOI: https://doi.org/10.1016/j.clinmicnews.2018.01.005
Mangold KA, Manson RU, Koay ES, et al. Real-time PCR for detection and identification of Plasmodium spp. J Clin Microbiol. 2005;43(5):2435-40. https://doi.org/10.1128/JCM.43.5.2435-2440.2005 DOI: https://doi.org/10.1128/JCM.43.5.2435-2440.2005
Beal SG, Assarzadegan N, Rand KH. Sample-to-result molecular infectious disease assays: clinical implications, limitations, and potential. Expert Rev Mol Diagn. 2016;16(3):323-41. https://pubmed.ncbi.nlm.nih.gov/26689497 https://pmc.ncbi.nlm.nih.gov/articles/PMC7103687 https://doi.org/10.1586/14737159.2016.1134325 DOI: https://doi.org/10.1586/14737159.2016.1134325
Suleiman M, Iqbal M, Tang P, Pérez-López A. Comparison of QIAstat-Dx and BioFire FilmArray gastrointestinal panels in a pediatric population. Microorganisms. 2024;12(11):2282. https://pubmed.ncbi.nlm.nih.gov/39597670 https://pmc.ncbi.nlm.nih.gov/articles/PMC11596080 https://doi.org/10.3390/microorganisms12112282 DOI: https://doi.org/10.3390/microorganisms12112282
Bispo PJM, Davoudi S, Sahm ML, et al. Rapid detection and identification of uveitis pathogens by qualitative multiplex real-time PCR. Invest Ophthalmol Vis Sci. 2018;59(1):582-9. Phttps://pubmed.ncbi.nlm.nih.gov/33584847 https://pmc.ncbi.nlm.nih.gov/articles/PMC7864790 https://doi.org/10.1016/j.nantod.2021.101092 DOI: https://doi.org/10.1167/iovs.17-22597
Wang C, Liu M, Wang Z, Li S, Dang Y, He N. Point-of-care diagnostics for infectious diseases: from methods to devices. Nano Today. 2021;37:101092. https://pubmed.ncbi.nlm.nih.gov/33584847 https://pmc.ncbi.nlm.nih.gov/articles/PMC7864790 https://doi.org/10.1016/j.nantod.2021.101092 DOI: https://doi.org/10.1016/j.nantod.2021.101092
Augustine R, Hasan A, Das S, et al. Loop-mediated isothermal amplification (LAMP): a rapid, sensitive, specific, and cost-effective point-of-care test for coronaviruses in the context of COVID-19 pandemic. Biology (Basel). 2020;9(8):182. https://pubmed.ncbi.nlm.nih.gov/32707972 https://pmc.ncbi.nlm.nih.gov/articles/PMC7464797 https://doi.org/10.3390/biology9080182 DOI: https://doi.org/10.3390/biology9080182
Garg N, Ahmad FJ, Kar S. Recent advances in loop-mediated isothermal amplification (LAMP) for rapid and efficient detection of pathogens. Curr Res Microb Sci. 2022;3:100120. https://pubmed.ncbi.nlm.nih.gov/35909594 https://pmc.ncbi.nlm.nih.gov/articles/PMC9325740 https://doi.org/10.1016/j.crmicr.2022.100120 DOI: https://doi.org/10.1016/j.crmicr.2022.100120
Hilt EE, Ferrieri P. Next generation and other sequencing technologies in diagnostic microbiology and infectious diseases. Genes (Basel). 2022;13(9):1566. https://pubmed.ncbi.nlm.nih.gov/36140733 https://pmc.ncbi.nlm.nih.gov/articles/PMC9498426 https://doi.org/10.3390/genes13091566 DOI: https://doi.org/10.3390/genes13091566
Chen Q, Yi J, Liu Y, et al. Clinical diagnostic value of targeted next generation sequencing for infectious diseases. Mol Med Rep. 2024;30(3):153. https://pubmed.ncbi.nlm.nih.gov/38963022 https://doi.org/10.3892/mmr.2024.13277 DOI: https://doi.org/10.3892/mmr.2024.13277
Park YE, Moon HS, Yong D, et al. Microbial changes in stool, saliva, serum, and urine before and after anti–TNF-α therapy in patients with inflammatory bowel diseases. Sci Rep. 2022;12(1):6359. https://pubmed.ncbi.nlm.nih.gov/35428806 https://pmc.ncbi.nlm.nih.gov/articles/PMC9012770 https://doi.org/10.1038/s41598-022-10450-2 DOI: https://doi.org/10.1038/s41598-022-10450-2
Quiñones-Mateu ME, Avila S, Reyes-Teran G, Martinez MA. Deep sequencing: becoming a critical tool in clinical virology. J Clin Virol. 2014;61(1):9-19. https://pubmed.ncbi.nlm.nih.gov/24998424 https://pmc.ncbi.nlm.nih.gov/articles/PMC4119849 https://doi.org/10.1016/j.jcv.2014.06.013 DOI: https://doi.org/10.1016/j.jcv.2014.06.013
Chen L, Cai Y, Zhou G, et al. Rapid Sanger sequencing of the 16S rRNA gene for identification of some common pathogens. PLoS One. 2014;9(2):e88886. https://pubmed.ncbi.nlm.nih.gov/24551186 https://pmc.ncbi.nlm.nih.gov/articles/PMC3925228 https://doi.org/10.1371/journal.pone.0088886 DOI: https://doi.org/10.1371/journal.pone.0088886
Tang W, Zhou Y, Li L, et al. Fasciola hepatica and Fasciola hybrid form co-existence in yak from Tibet of China: application of rDNA internal transcribed spacer. Parasitol Res. 2024;123(11):366. https://pubmed.ncbi.nlm.nih.gov/39482528 https://doi.org/10.1007/s00436-024-08383-y DOI: https://doi.org/10.1007/s00436-024-08383-y
Lavezzo E, Barzon L, Toppo S, Palù G. Third generation sequencing technologies in diagnostic microbiology: benefits and challenges. Expert Rev Mol Diagn. 2016;16(9):1011-23. https://pubmed.ncbi.nlm.nih.gov/27453996 https://doi.org/10.1080/14737159.2016.1217158 DOI: https://doi.org/10.1080/14737159.2016.1217158
Schmidt J, Blessing F, Fimpler L, Wenzel F. Nanopore sequencing in a clinical routine laboratory: challenges and opportunities. Clin Lab. 2020;66(6). https://pubmed.ncbi.nlm.nih.gov/32538066 https://doi.org/10.7754/Clin.Lab.2019.191114 DOI: https://doi.org/10.7754/Clin.Lab.2019.191114
Kunasol C, Dondorp AM, Batty EM, et al. Comparative analysis of targeted next-generation sequencing for Plasmodium falciparum drug resistance markers. Sci Rep. 2022;12(1):5563. https://pubmed.ncbi.nlm.nih.gov/35365711 https://pmc.ncbi.nlm.nih.gov/articles/PMC8974807 https://doi.org/10.1038/s41598-022-09474-5 DOI: https://doi.org/10.1038/s41598-022-09474-5
Nicot F, Trémeaux P, Latour J, et al. Whole-genome single molecule real-time sequencing of SARS-CoV-2 Omicron. J Med Virol. 2023;95(2):e28564. https://pubmed.ncbi.nlm.nih.gov/36756931 https://doi.org/10.1002/jmv.28564 DOI: https://doi.org/10.1002/jmv.28564
Raymond S, Jeanne N, Vellas C, et al. HIV-1 genotypic resistance testing using single molecule real-time sequencing. J Clin Virol. 2024;174:105717. https://pubmed.ncbi.nlm.nih.gov/39068746 https://doi.org/10.1016/j.jcv.2024.105717 DOI: https://doi.org/10.1016/j.jcv.2024.105717
Simner PJ, Miller S, Carroll KC. Understanding the promises and hurdles of metagenomic next-generation sequencing as a diagnostic tool for infectious diseases. Clin Infect Dis. 2018;66(5):778-88. https://pubmed.ncbi.nlm.nih.gov/29040428 https://pmc.ncbi.nlm.nih.gov/articles/PMC7108102 https://doi.org/10.1093/cid/cix881 DOI: https://doi.org/10.1093/cid/cix881
Ko KKK, Chng KR, Nagarajan N. Metagenomics-enabled microbial surveillance. Nat Microbiol. 2022;7(4):486-96. https://pubmed.ncbi.nlm.nih.gov/35365786 https://doi.org/10.1038/s41564-022-01089-w DOI: https://doi.org/10.1038/s41564-022-01089-w
Gu W, Deng X, Lee M, et al. Rapid pathogen detection by metagenomic next-generation sequencing of infected body fluids. Nat Med. 2021;27(1):115-24. https://pubmed.ncbi.nlm.nih.gov/33169017 https://pmc.ncbi.nlm.nih.gov/articles/PMC9020267 https://doi.org/10.1038/s41591-020-1105-z DOI: https://doi.org/10.1038/s41591-020-1105-z
Wani AK, Chopra C, Dhanjal DS, et al. Metagenomics in the fight against zoonotic viral infections: a focus on SARS-CoV-2 analogues. J Virol Methods. 2024;323:114837. https://pubmed.ncbi.nlm.nih.gov/37914040 https://doi.org/10.1016/j.jviromet.2023.114837 DOI: https://doi.org/10.1016/j.jviromet.2023.114837
Wang D, Coscoy L, Zylberberg M, et al. Microarray-based detection and genotyping of viral pathogens. Proc Natl Acad Sci U S A. 2002;99(24):15687-92. https://pubmed.ncbi.nlm.nih.gov/12429852 https://pmc.ncbi.nlm.nih.gov/articles/PMC137777 https://doi.org/10.1073/pnas.242579699 DOI: https://doi.org/10.1073/pnas.242579699
Ma X, Li Y, Liang Y, et al. Development of a DNA microarray assay for rapid detection of fifteen bacterial pathogens in pneumonia. BMC Microbiol. 2020;20(1):177. https://pubmed.ncbi.nlm.nih.gov/32576241 https://pmc.ncbi.nlm.nih.gov/articles/PMC7310556 https://doi.org/10.1186/s12866-020-01842-3 DOI: https://doi.org/10.1186/s12866-020-01842-3
Krishnamurthy HK, Jayaraman V, Krishna K, et al. A customizable multiplex protein microarray for antibody testing and its application for tick-borne and other infectious diseases. Sci Rep. 2025;15(1):2527. Phttps://pubmed.ncbi.nlm.nih.gov/39833196 https://pmc.ncbi.nlm.nih.gov/articles/PMC11747503 https://doi.org/10.1038/s41598-024-84467-0 DOI: https://doi.org/10.1038/s41598-024-84467-0
Gavina K, Franco LC, Khan H, Lavik JP, Relich RF. Molecular point-of-care devices for the diagnosis of infectious diseases in resource-limited settings. J Clin Virol. 2023;169:105613. https://pubmed.ncbi.nlm.nih.gov/37866094 https://doi.org/10.1016/j.jcv.2023.105613 DOI: https://doi.org/10.1016/j.jcv.2023.105613
Najjar D, Rainbow J, Sharma Timilsina S, et al. A lab-on-a-chip for concurrent electrochemical detection of SARS-CoV-2 RNA and antibodies in saliva and plasma. Nat Biomed Eng. 2022;6(8):968-78. https://pubmed.ncbi.nlm.nih.gov/35941191 https://pmc.ncbi.nlm.nih.gov/articles/PMC9361916 https://doi.org/10.1038/s41551-022-00919-w DOI: https://doi.org/10.1038/s41551-022-00919-w
Padmanaban V, Ranganathan UDK. CRISPR-Cas system and its use in the diagnosis of infectious diseases. Microbiol Res. 2022;263:127100. https://pubmed.ncbi.nlm.nih.gov/35849921 https://doi.org/10.1016/j.micres.2022.127100 DOI: https://doi.org/10.1016/j.micres.2022.127100
Broughton JP, Deng X, Yu G, et al. CRISPR-Cas12–based detection of SARS-CoV-2. Nat Biotechnol. 2020;38(7):870-4. https://pubmed.ncbi.nlm.nih.gov/32300245 https://pmc.ncbi.nlm.nih.gov/articles/PMC9107629 https://doi.org/10.1038/s41587-020-0513-4 DOI: https://doi.org/10.1038/s41587-020-0513-4
Kellner MJ, Koob JG, Gootenberg JS, Abudayyeh OO, Zhang F. SHERLOCK: nucleic acid detection with CRISPR nucleases. Nat Protoc. 2019;14(10):2986-3012. https://pubmed.ncbi.nlm.nih.gov/ 31548639 https://pmc.ncbi.nlm.nih.gov/articles/PMC6956564 https://doi.org/10.1038/s41596-019-0210-2 DOI: https://doi.org/10.1038/s41596-019-0210-2
Wu Y, Battalapalli D, Hakeem MJ, et al. Engineered CRISPR-Cas systems for the detection and control of antibiotic-resistant infections. J Nanobiotechnology. 2021;19(1):401. https://pubmed.ncbi.nlm.nih.gov/34863214 https://pmc.ncbi.nlm.nih.gov/articles/PMC8642896 https://doi.org/10.1186/s12951-021-01132-8 DOI: https://doi.org/10.1186/s12951-021-01132-8
Myhrvold C, Freije CA, Gootenberg JS, et al. Field-deployable viral diagnostics using CRISPR-Cas13. Science. 2018;360(6387):444-8. https://pubmed.ncbi.nlm.nih.gov/29700266 https://pmc.ncbi.nlm.nih.gov/articles/PMC6197056 https://doi.org/10.1126/science.aas8836 DOI: https://doi.org/10.1126/science.aas8836
Fozouni P, Son S, Díaz de León Derby M, et al. Amplification-free detection of SARS-CoV-2 with CRISPR-Cas13a and mobile phone microscopy. Cell. 2021;184(2):323-33.e9. https://pubmed.ncbi.nlm.nih.gov/33306959 https://pmc.ncbi.nlm.nih.gov/articles/PMC7834310 https://doi.org/10.1016/j.cell.2020.12.001 DOI: https://doi.org/10.1016/j.cell.2020.12.001
Baylis SA, Wallace P, McCulloch E, Niesters HGM, Nübling CM. Standardization of nucleic acid tests: the approach of the World Health Organization. J Clin Microbiol. 2019;57(1):e01056-18. https://pubmed.ncbi.nlm.nih.gov/30257900 https://pmc.ncbi.nlm.nih.gov/articles/PMC6322456 https://doi.org/10.1128/JCM.01056-18 DOI: https://doi.org/10.1128/JCM.01056-18
Wong F, de la Fuente-Nunez C, Collins JJ. Leveraging artificial intelligence in the fight against infectious diseases. Science. 2023;381(6654):164-70. https://pubmed.ncbi.nlm.nih.gov/29700266 https://pmc.ncbi.nlm.nih.gov/articles/PMC6197056 https://doi.org/10.1126/science.aas8836 DOI: https://doi.org/10.1126/science.adh1114
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Michael Baclig

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.









 @philippinepathologyjournal
@philippinepathologyjournal