Determination of Rates of Malignancy on Archival Salivary Gland Fine-Needle Aspiration Biopsy after Application of the Milan System for Reporting Salivary Gland Cytopathology in the Philippine General Hospital
A 1-Year Retrospective Study
DOI:
https://doi.org/10.21141/PJP.2022.09Keywords:
cytopathology, fine needle aspiration biopsy, FNAB, Milan System, salivary gland, rates of malignancyAbstract
Background. The Milan System for Reporting Salivary Gland Cytopathology (MSRGC) aims to increase the overall effectiveness of salivary gland FNAB by defining six general diagnostic categories with corresponding Rates of Malignancies (ROM). This study aims to use this system to categorize salivary gland FNAB in the Philippine General Hospital and stratify ROM per category.
Methodology. In this study a total of 326 cases have been collected and reviewed, of which 154 (47.2%) had either surgical or clinical follow-up. The cases were assigned a Milan category by 3 cytopathologists blinded from the original diagnoses and from each other’s readings.
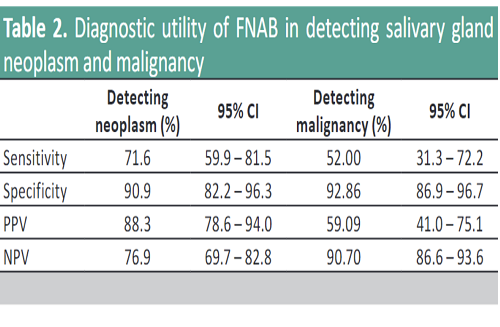
Results. The overall sensitivity, specificity, PPV, and NPV in detecting neoplasm is at 71.6%, 90.9%, 88.3%, and 76.9%, respectively. On the other hand, the sensitivity, specificity, PPV, and NPV in detecting malignancy is at 52%, 92.9%, 59.1%, and 90.7%, respectively. The computed ROM is as follows: Category I 7.89%, Category II 9.43%, Category III 20%, Category IVa 10.53%, Category IVb 60%, Category V 75%, and Category VI 100%.
Conclusion. The overall diagnostic utility of salivary gland FNAB, as well as the computed ROM per diagnostic category are comparable to internationally published literature. This study also validates the MSRSGC as a valuable tool in stratifying ROM in salivary gland lesions.
Downloads
References
2. Rossi ED, Wong, LQ, Bizzarro T, et al. The impact of FNAC in the management of salivary gland lesions: institutional experiences leading to a risk-based classification scheme. Cancer Cytopathol. 2016;124(6):388-96. https://pubmed.ncbi.nlm.nih.gov/26959289. https://doi.org/10.1002/cncy.21710.
3. Faquin WC and Rossi ED (eds). The Milan System for reporting salivary gland cytology. Cham, Springer; 2018.
4. Griffith CC, Pai RK, Schneider F, et al. Salivary gland tumor fine-needle aspiration cytology: a proposal for a risk stratification classification. Am J Clin Pathol. 2015;143(6): 839-53. https://pubmed.ncbi.nlm.nih.gov/25972326. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5257286. https://doi.org/10.1309/AJCPMII6OSD2HSJA..
5. Vallonthaiel AG, Kaushal S, Jangir H, Rajendran HK. Application of the Milan system for risk stratification and its comparison with a previous reporting system of parotid gland cytopathology in a tertiary care center. Acta Cytologica. 2018;62(5-6):352-9. https://pubmed.ncbi.nlm.nih.gov/30223278. https://doi.org10.1159/000492051.
6. Rossi ED, Faquin WC, Zubair B, et al. The Milan system for reporting salivary gland cytopathology: analysis and suggestions of initial survey. Cancer Cytopathol. 2017;125(10):757-66. https://pubmed.ncbi.nlm.nih.gov/28708928. https://doi.org/10.1002/cncy.21898.
7. Rossi ED, Baloch ZQ, Pusztaszeri M, Faquin W. The Milan system for reporting salivary gland cytopathology (MSRSGC): an ASC-IAC-sponsored system for reporting salivary gland fine-needle aspiration. J Am Soc Cytopathol. 2018;7(3):111-8. https://pubmed.ncbi.nlm.nih.gov/31043307. https://doi.org/10.1016/j.jasc.2018.02.002.
8. Kessler AT, Bhatt AA. Review of the major and minor salivary glands, part 1: anatomy, infectious, and inflammatory processes. J Clin Imaging Sci. 2018;8:47. https://pubmed.ncbi.nlm.nih.gov/30546931. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6251248. https://doi.org/10.4103/jcis.JCIS_45_18.
9. Hang JF, Alruwaii F, Zeng BR, Lai CR, Wu HH. Subtyping salivary gland neoplasm of uncertain malignant potential based on cell type demonstrates differential risk of malignancy. Cancer Cytopathol. 2018;126(11):924-33. https://pubmed.ncbi.nlm.nih.gov/30335220. https://doi.org/10.1002/cncy.22066.
10. Zhang S, Bao R, Bagby J, Abreo F. Fine needle aspiration of salivary glands: 5-year experience from a single academic center. Acta Cytologica. 2009;53(4):375-82. https://pubmed.ncbi.nlm.nih.gov/19697720. https://doi.org/10.1159/000325336.
11. Al-Khafaji BM, Nestok BR, Katz RL. Fine-needle aspiration of 154 parotid masses with histologic correlation: ten-year experience at the University of Texas M.D. Anderson Cancer Center. Cancer. 1998;84(3):153-9. https://pubmed.ncbi.nlm.nih.gov/9678729.
12. Stewart CJ, MacKenzie K, McGarry GW, Mowat A. Fine-needle aspiration cytology of salivary gland: a review of 341 cases. Diagn Cytopathol. 2000;22(3):139-46. https://pubmed.ncbi.nlm.nih.gov/10679992. https://doi.org/10.1002/(sici)1097-0339(20000301)22:3<139::aid-dc2>3.0.co;2-a.
13. Wang H, Malik A, Maleki Z, et al. Atypical salivary gland fine needle aspiration: risk of malignancy and inter-institutional variability. Diagn Cytopathol. 2017;45(12):1088-94. https://pubmed.ncbi.nlm.nih.gov/28960946. https://doi.org/10.1002/dc.23826.
14. Madani G. Imaging of salivary glands. In: Maxillofacial Surgery, 3rd ed. 2017. https://doi.org/10.1016/B978-0-7020-6056-4.00048-4.
15. Ameli F, Baharoom A, Md Isa N, Akmal SN. Diagnostic challenges in fine needle aspiration cytology of salivary gland lesions. Malaysian J Pathol. 2015;37(1):11-8. https://pubmed.ncbi.nlm.nih.gov/25890608.
16. Frable MA, Frable WJ. Fine needle aspiration biopsy of salivary glands. Laryngoscope. 1991; 101(3):245-9. https://pubmed.ncbi.nlm.nih.gov/2000011. https://doi.org/10.1288/00005537-199103000-00005.
17. Ersöz C, Uguz A, Tuncer Ü, Soylu L. Fine needle aspiration cytology of the salivary glands: a twelve years’ experience. Aegean Pathol J. 2004;1:51-6.
18. Wei S, Layfield LJ, LiVolsi VA, Montone KT, Baloch ZW. Reporting of fine needle aspiration (FNA) specimens of salivary gland lesions: a comprehensive review. Diagn Cytopathol. 2017; 45(9):820-7. https://pubmed.ncbi.nlm.nih.gov/28371507. https://doi.org/10.1002/dc.23716.
19. Santiago KJB, Roldan RA, Castañeda SS. Accuracy of fine needle aspiration biopsy in diagnosing parotid gland malignancy. Philipp J Otorlaryngol Head Neck Surg. 2016;31(2):24-6. https://doi.org/10.32412/pjohns.v31i2.229.
20. Schmidt RL, Hall BJ, Wilson Ar, Layfield LJ. A systematic review and meta-analysis of the diagnostic accuracy of fine-needle aspiration cytology for parotid gland lesions. Am J Clin Pathol. 2017;136(1):45-59. https://pubmed.ncbi.nlm.nih.gov/21685031. https://doi.org/10.1309/AJCPOIE0CZNAT6SQ.
21. Inançlı HM, Kanmaz MA, Ural A, Dilek GB. Fine needle aspiration biopsy: in the diagnosis of salivary gland neoplasms compared with histopathology. Indian J Otolaryngol Head Neck Surg. 2013;65 (Suppl 1):121-5. https://pubmed.ncbi.nlm.nih.gov/24427627. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3718948. https://doi.org/10.1007/s12070-012-0608-4.
22. Layfield LJ, Glasgow BJ. Diagnosis of salivary gland tumors by fine needle aspiration cytology: A review of clinical utility and pitfalls. Diagn Cytopathol. 1991;7(3):267–72. https://pubmed.ncbi.nlm.nih.gov/1879262. https://doi.org/10.1002/dc.2840070311.
23. O’dwyer P, Farrar WB, James AG, Finkelmeier W, McCabe DP. Needle aspiration biopsy of major salivary gland tumors. Cancer 1986;57(3):554-7. https://pubmed.ncbi.nlm.nih.gov/3942989. https://doi.org/10.1002/1097-0142(19860201)57:3<554::aid-cncr2820570325>3.0.co;2-g.
24. Viswanathan K, Sung S, Scognamiglio T, Yang GCH, Siddiqui MT, Rao RA. The role of the Milan system for reporting salivary gland cytopathology: a 5-year institutional experience. Cancer Cytopathol. 2018;126(8): 541-51. https://pubmed.ncbi.nlm.nih.gov/29797690. https://doi.org/10.1002/cncy.22016.
25. Qizilbash AH, Sianos J, Young JE, Archibald SD. Fine needle aspiration biopsy cytology of major salivary glands. Acta Cytol. 1985;29(4):503-12. https://pubmed.ncbi.nlm.nih.gov/2992196.
26. Young JE, Archibald SD, Shier KJ. Needle aspiration cytologic biopsy in head and neck masses. Am J Surg 1981;142(4):484-9. https://pubmed.ncbi.nlm.nih.gov/7283052. https://doi.org/10.1016/0002-9610(81)90380-9.
27. Thiryayi SA, Low YX, Shelton D, Narine N, Slater D, Rana DN. A retrospective three-year study of salivary gland fine needle aspiration cytology with categorization using the Milan reporting system. Cytopathology. 2018;29(4):343-8. https://pubmed.ncbi.nlm.nih.gov/29683536. https://doi.org/10.1111/cyt.12557.
28. Kala C, Kala S, Khan L. Milan system for reporting salivary gland cytopathology: an experience with the implication for risk of malignancy. J Cytol. 2019;36(3):160-4. https://pubmed.ncbi.nlm.nih.gov/31359916. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6592120. https://doi.org/10.4103/JOC.JOC_165_18.
29. Liang CA, Liu J, Ogunniyi JT, Zhu H, Songlin Z. The risk for malignancy using the Milan salivary gland classification categories: a 5-year retrospective review. Cytojournal. 2019;16:14. https://pubmed.ncbi.nlm.nih.gov/31516536. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6683416. https://doi.org/10.4103/cytojournal.cytojournal_45_18.









 @philippinepathologyjournal
@philippinepathologyjournal